A 'Pandemic' of Sloppy COVID-19 PCR Testing Produced Countless False Positives and a Highly Inaccurate Picture of the True Number of Cases
A Scientific Explanation of the Fraud of RT-PCR Testing for COVID-19
“The COVID-19 PCR tests were a fraud.” You have heard this before, but is it true or just more hype? Read this report and decide for yourself. It explains the fundamentals of the RT-PCR (Reverse Transcriptase - Polymerase Chain Reaction) test kits for those who want to understand the facts. Do not expect an easy read. For starters, you will need to familiarize yourself with the basic structure of DNA. Understanding how misinterpretation of the RT-PCR test results was used to create the COVID-19 pandemic will be your reward. You will be immunized against future efforts to create fear and societal discord. Rest assured; they will try again!
A serious flu-like respiratory disease began to spread in early Fall 2019 with Wuhan, China, as “ground zero.” By December, worldwide spread of the disease was underway. Images of hapless pedestrians suddenly falling dead in the streets; overwhelmed hospitals in Lombardy, Italy; government-imposed lockdowns; and nonstop coverage of worldwide COVID deaths fanned the flames of fear surrounding this unknown disease. It was not enough to be symptom-free. People demanded the development of a test to detect silent carriers of the infectious agent.
Only the genome sequence of SARS-CoV-2 was known at the time, and the only available testing method used polymerase chain reaction, a.k.a. PCR. Despite many known contraindications, RT-PCR widespread testing for COVID-19 began. According to Dr. Trish M. Perl, an epidemiologist at Johns Hopkins and past president of the Society of Health Care Epidemiologists of America, blind faith in the results of technically complex molecular tests results in “pseudo-epidemics.” [https://www.nytimes.com/2007/01/22/health/22whoop.html] The COVID-19 pandemic was one of those.
Even Kary Mullis, the Nobel Prize-winning inventor of PCR, would not have approved. PCR revolutionized the life sciences, especially molecular biology. This extraordinary biochemical tool facilitated all manner of techniques that were close to impossible without it. Unfortunately, demonstrating the presence of a VIABLE infectious agent is not within PCR’s skill set. Although Mullis was talking about HIV, the following quote is still appropriate for COVID-19. “[PCR] doesn’t tell you that you are sick, or that the thing that you ended up with was going to hurt you or anything like that.” Mullis continues, “It allows you to take a minuscule amount of anything and make it measurable and then talk about it.” [https://off-guardian.org/2020/10/05/pcr-inventor-it-doesnt-tell-you-that-you-are-sick/]
In the end, it was not the PCR technique that was the problem, but the fraudulent analysis of the results. To quote Mullis, “I don’t think you can misuse PCR. [It is] the results; the interpretation of it. If they can find this virus in you at all – and with PCR, if you do it well, you can find almost anything in anybody.” [https://off-guardian.org/2020/10/05/pcr-inventor-it-doesnt-tell-you-that-you-are-sick/] The FDA, which approved commercial RT-PCR test kits for COVID-19 and developed the first kits, should have been aware of the potential for misuse. It was their responsibility to put in place guardrails to prevent misuse. The CDC should have been informed as well. The fact that the FDA kept silent, or allowed it to happen, speaks volumes.
What is PCR?
PCR is a molecular copy machine capable of producing trillions of copies of a short section of a DNA molecule. It relies on features of the DNA synthesis machinery revealed by decades of research, for which many have received Nobel Prizes.
PCR uses a special thermostable DNA polymerase, the enzyme that synthesizes DNA, that can withstand heating to temperatures of 92oC to 98o
DNA polymerase makes copies of a “template” DNA molecule. It requires a template, and it cannot start synthesizing DNA just anywhere along a DNA template. It moves along one strand of the DNA template, adding one nucleotide (base) at a time. The newly inserted nucleotide base is “complementary” to the base on the template strand. Complementary pairs of nucleotides are as follows. A is complementary to T, and G is complementary to C.
Single strands of DNA have chemically distinct ends referred to as the 5’ and 3’ ends. DNA polymerase can only synthesize the new DNA strand by attaching to a free 3’ end. In PCR, the free 3’ end is provided by a short single strand of nucleotides, about 12 to 20 nucleotides in length, called a “primer.” DNA polymerase starts synthesizing DNA by attaching to the 3’-end of the primer.
PCR uses pairs of primers to make multiple copies of the section of DNA between the primers. The two primers define the ends of the section to be copied. The sequence of the primers must be carefully determined to be complementary to unique sequences on opposite strands of the template DNA. Recognition of unique sequences by the primers is how PCR distinguishes among the other closely related human coronaviruses endemic in the population and SARS-CoV-2.
At 37oC to 65oC, primers pair up with their complementary sequence, a process called “annealing.” The accuracy and stringency of primer annealing are dependent on the length of the primer, its G/C content, and the annealing temperature used. The higher the annealing temperature, the more stringent the sequence complementarity required between the primer and the target site. The proper annealing temperature for high stringency pairing is easily calculated for each primer.
At temperatures of 92oC to 98oC, double-stranded DNA separates into single strands, called “melting.”
DNA synthesis starting at the 3’end of the primer is called “primer extension” and occurs at 72o.
A typical PCR reaction mix contains (1) thermostable DNA polymerase that can withstand temperatures up to 92oC to 98oC; (2) the pair of primers; (3) the target DNA; (4) all four nucleotides (A, T, C, G) bases, and (5) buffers and salt solutions. The PCR reaction mix is placed in a very small, conical-bottom plastic tube capable of holding only 10 or fewer drops of reaction mix.
This tube is inserted into the thermoblock of a PCR thermocycler. A typical thermoblock is about 7 centimeters square and one to two centimeters thick. It contains multiple “wells,” holes whose size perfectly fit the conical-bottom plastic tubes used for the PCR reactions. A typical number of wells is 96 arranged in eight rows of 12. The thermoblock is placed into the PCR thermocycler “temperature bath,” and all of the tubes are sealed in place.
The thermocycler rapidly raises and lowers the temperature of the thermoblock/reaction tubes in a matter of seconds. This is computer-controlled and is input by the technician. Temperature and times of each cycle will vary. Each PCR cycle consists of the following series of temperature changes and takes only a few minutes.
The temperature is raised to allow all of the DNA strands to separate into single strands.
The temperature is lowered to the desired annealing temperature to allow the primers to pair to their unique complementary sequence.
The temperature is raised to 72oC to allow the thermostable DNA polymerase to extend the primer and produce a strand of DNA complementary to the target DNA template.
Reverse Transcriptase
SARS-CoV-2 is a single-stranded RNA that functions as a messenger RNA (mRNA) for protein synthesis when it infects a cell. PCR needs a DNA target. Thus, to use PCR to detect the presence of SARS-CoV-2, this RNA must first be copied into DNA. This is done using a special DNA polymerase called “reverse transcriptase” which uses RNA as the template to synthesize a complementary DNA strand.
RT-PCR Test Procedure
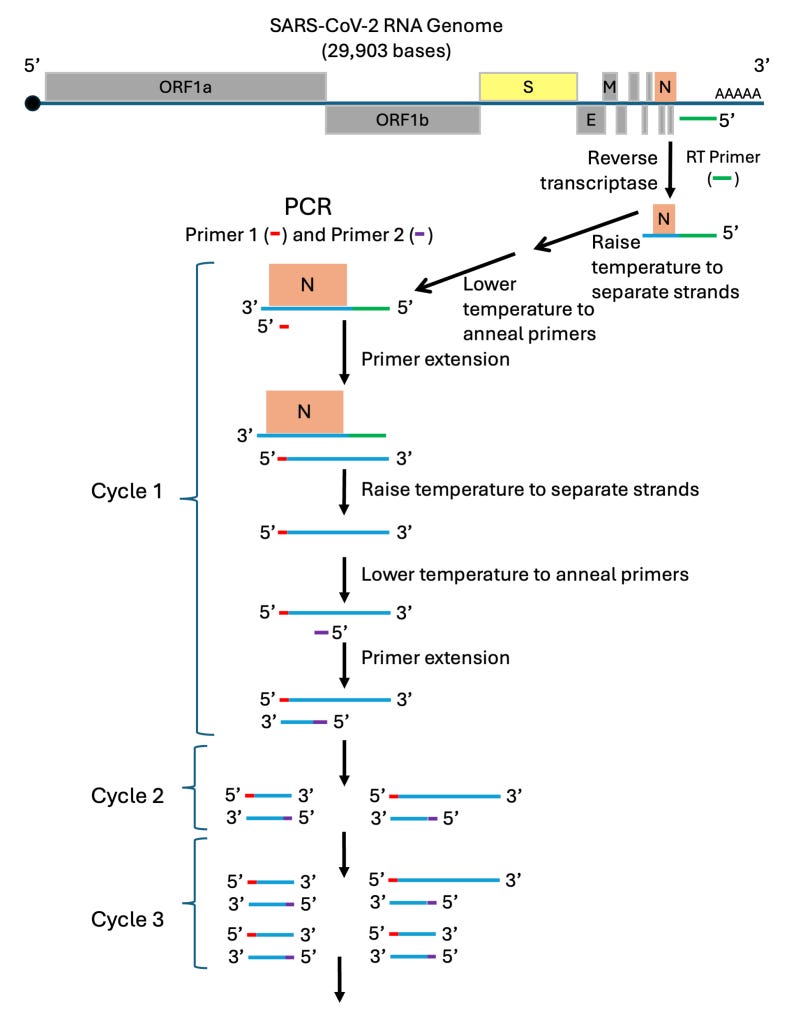
Step 1: Extract RNA from the nasal swab. This will include any SARS-CoV-2 RNA plus host RNA and other nasal contaminants.
Step 2: Reverse transcriptase is used to synthesize a DNA complement to a specific section of SARS-CoV-2 RNA. In the diagram, the RT-primer for this step (in green) is complementary to a unique sequence in the untranslated region found at the 3’-end of SARS-CoV-2, just next to the N gene. The DNA strand produced will only extend a few hundred bases into the N gene. It will not include the Spike gene or any other genes of this almost 30,000-nucleotide-long genome. This is an important point! Only the presence of the N gene is being tested. (Other regions of the SARS-CoV-2 RNA could be tested as long as a unique RT-primer sequence is used.)
Step 3: Raise the temperature to separate the short DNA strand from the SARS-CoV-2 RNA.
Step 4: Lower the temperature to allow Primer 1 (in red) of the PCR mix to pair up (anneal) to the N gene DNA strand. Primer 1 is complementary to a unique short sequence that is inside the boundary of the N gene.
Step 5: Raise the temperature to about 72oC to allow the temperature-stable DNA polymerase to synthesize the DNA complementary strand, called primer extension.
Step 6: Raise the temperature to separate the DNA strands.
As long as the primers used pair with sequences that are unique to SARS-CoV-2 RNA, the RT-PCR test will detect only SARS-CoV-2 RNA. If, for any reason, the sequence of the primers is incorrectly decided upon, the test will be flawed and give incorrect results. [Borger, Pieter, Malhotra, Rajesh Kumar, Yeadon, Michael, Craig, Clare, McKernan, Kevin, Steger, Klaus, … Kämmer, Ulrike. (2020). External peer review of the RTPCR test to detect SARS-CoV-2 reveals 10 major scientific flaws at the molecular and methodological level: consequences for false positive results. http://doi.org/10.5281/zenodo.4298004]
Step 1 produces an RNA/DNA hybrid of the N gene section of SARS-CoV-2 RNA. PCR cycle 1 produces one double-stranded fragment of DNA copied from the region between the primer pair. Cycle 2 produces two copies of this fragment; Cycle 3 produces four copies; and so forth. The amount of double-stranded DNA produced is quantified easily using different methods and can be done in the heating blocks real-time thermocyclers.
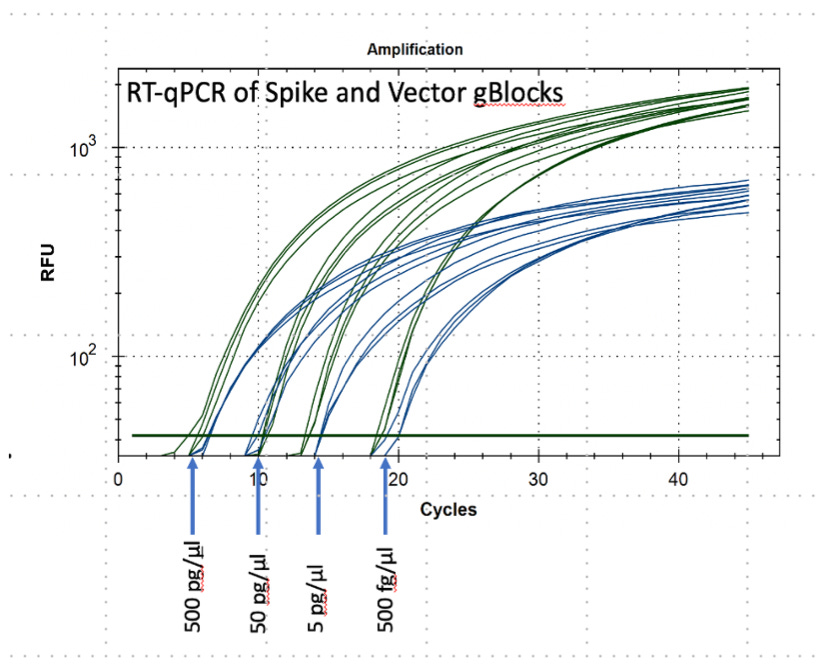
Based on one starting copy, 25 cycles will produce 335,543,936 copies, and 45 cycles will produce 3,474,749,302,776 copies. If there is more than one copy in the starting reaction mix, these numbers will be reached after fewer cycles. In other words, the fewer number of cycles needed to detect a fragment, the higher the number of input copies.
DNA-binding “probes” release a fluorescent dye molecule each time a DNA fragment is copied. When a minimum number of dye molecules are released, the PCR reaction mix begins to fluoresce. The florescence of each reaction tube is monitored by the thermocycler, and the number of cycles is recorded. See a typical plot of RT-PCR results.
Where RT-PCR Testing Went Wrong
PCR requires specialized equipment and training not only to perform the procedures properly but also to analyze the results correctly. By February 3, 2020, the U.S. Centers for Disease Control and Prevention (CDC) had developed the first RT-PCR test kit. Dozens of other commercially produced RT-PCR testing kits were soon developed. So, what could go wrong? Tons of things! Just a few of the most significant are listed below. [https://dailysceptic.org/2020/11/30/the-pcr-false-positive-pseudo-epidemic/]
As stated in the CDC’s instruction document for its RT-PCR test kit, [it] is intended for use by laboratories designated by CDC as qualified and “to be limited to laboratories certified under the Clinical Laboratory Improvement Amendments (CLIA) that meet the requirements to perform high complexity tests”. [https://www.fda.gov/media/134922/download] This, of course, was true for all of the RT-PCR testing kits. Billions of PCR tests were run at tens of thousands of sites, most of which were not CLIA-certified. This is comparable to taking your Ferrari 12 Cilindri for an engine tune-up by your local garage mechanic. The explosion of the number of testing sites increased the probability of testing mistakes.
The most important take-home message from the diagram above is that the RT-PCR test does not discriminate between the whole virus and fragments of the viral RNA genome. It is ONLY capable of demonstrating the presence of SARS-CoV-2 fragments. It cannot be used as an indicator of the presence of infectious (encapsulated) viral particles in the swab. Misinterpretation of RT-PCR test results explains the “asymptomatic carrier,” the claims that so many people hospitalized for other reasons were declared to have COVID-19, and why so much of the lockdown frenzy we went through was entirely needless. There was a “pandemic of testing!”
Patient sampling errors due to variations in “swabbing techniques” and in the efficiency of RNA purification techniques can lead to 1,000 to 10,000-fold differences in the amount of RNA in the sample tested. This translates to differences of up to 10 to 13 PCR cycles. [Michael Yeadon, “The PCR False Positive Pseudo-Epidemic.” https://lockdownsceptics.org/the-pcr-false-positive-pseudo-epidemic/]
Test protocols should have included positive and negative control samples in each sample series. These controls would serve as indicators of technique errors and contamination. RNA is easily degraded, and poor extraction methods lead to false negative results. Contamination is a major problem in PCR because of its extreme sensitivity and sloppy technique can lead to false positive results. [https://www.thelancet.com/action/showPdf?pii=S2213-2600%2820%2930453-7]
A significant source of analysis error in the RT-PCR test was the omission of a cut-off number of cycles at which a sample was to be considered positive or negative. Documentation for the testing kits referred to the RT-PCR test as “qualitative.” The simple addition of a standardized sample carrying a known number of copies of the DNA target would have easily allowed quantification. This is obvious from the dilution series shown in the Amplification figure above. If this had been done, a cut-off of about 25 cycles would have been sufficient to demonstrate a viral load sufficient to indicate a true infection. Testing centers rarely reported the number of cycles they were using to call a sample “positive,” but reports indicated that 45 to 47 cycles were commonly used. This number of cycles is capable of detecting insignificant nasal contaminants rather than a true infection. [https://www.journalofinfection.com/action/showPdf?pii=S0163-4453%2820%2930675-7]
Extremely high concentrations of the primers were used in some kit protocols, which could have contributed to aberrant annealing to sites in coronavirus strains other than SARS-CoV-2.
The annealing temperature for a primer pair is critical and must be carefully set. Different RT-PCR kits from different commercial providers used different primer pairs and, thus, would require different annealing temperatures. If a testing site used different commercial kits, it would have been critical that the thermocycler equipment temperatures be appropriately set for each kit type.
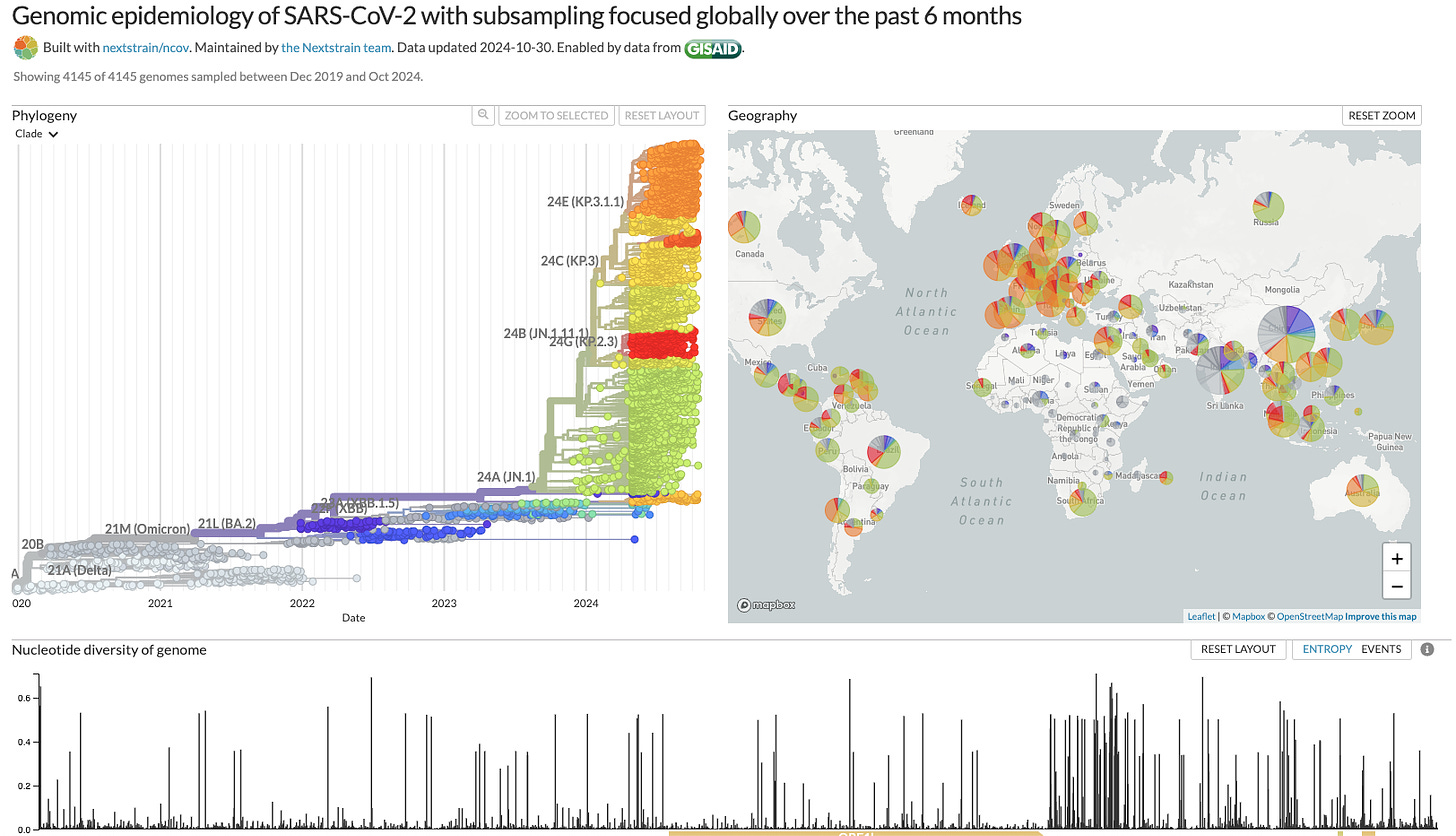
SARS-CoV-2 spread was accompanied by accelerated production of hundreds of variants. The multitude of sequence changes accumulated throughout the length of the viral genome including the N gene. For a visualization of this, see the screenshot below from the NextStrain site entitled, “Genomic epidemiology of SARS-CoV-2 with subsampling focused globally over the past 6 months” (https://nextstrain.org/ncov/gisaid/global/6m)
To keep pace with constant sequence variation, continued updating of the primer sets for a particular RT-PCR kit is necessary. Some but not all commercial kits were updated. Without appropriately updated primers, the frequency of false negative test results would increase, and individuals sick with a new SARS2-CoV-2 variant would be misdiagnosed by the reputed “gold standard” test for COVID-19.
In November 2023, the Acon Flowflex COVID-19 Antigen Home Test became the first of dozens of at-home rapid antigen tests approved by the U.S. Food and Drug Administration (FDA) for identifying COVID-19 infection. Antigen test kits detect the presence of SARS-CoV-2 N capsid protein, an essential protein involved in packaging the viral RNA into the virus particle. These tests are used in the first one to five days of symptoms and are clear indicators of an active infection.
The availability of antigen home tests ended the frenzy of RT-PCR testing for the general public. More importantly, they ushered in the end of the pandemic frenzy and “contact tracing” frenzy. Hospitals and doctor offices continued to use the RT-PCR test, likely because administrators demanded this cash cow of government money.
RT-PCR testing for COVID-19 infection was a monstrous fraud imposed on a trusting populous. The COVID pandemic was really the Pandemic of Testing! It was used to lock Americans down, isolate us from friends and family, create rampant fear, and make us willing to vaccinate with an experimental vaccine. Government and public health entities succeeded once because the public was naive. They will continue to use the same ploy with the newest flavor of the month viral disease. Next time, reason, not irrational fear, will keep us free.
Corinne Michels, PhD, is an internationally recognized molecular geneticist studying the regulation of gene expression in the model eukaryote Saccharomyces cerevisiae, baker’s and brewer’s yeast. Her research was consistently funded by grants from the US National Institutes of Health and the National Science Foundation. She authored of over 75 research journal articles, review articles, and book chapters in scientific publications. Dr. Michels’ textbook, Genetic Techniques for Biological Research: A Case Study Approach (John Wiley & Sons), is widely used in both the classroom and research laboratory.
This article is the writer’s opinion. Freedom of speech is one of America’s most important freedoms.




Anyone that took this tests believing they were legit is the first problem. I never took one and knew rhey were BS day 1. The guy that designed them said it was BS and then was dead a month later. It's a worldwide scam
Lord give us all increased wisdom & strength with His Power!